Abstract
In the struggle between virus and host, control over the cell's death machinery is crucial for survival. Viruses are obligatory intracellular parasites and, as such, must modulate apoptotic pathways to control the lifespan of their host in order to complete their replication cycle. Many of the counter-assaults mounted by the immune system incorporate activation of the apoptotic pathway—particularly by members of the tumor necrosis factor cytokine family—as a mechanism to restrict viral replication. Thus, apoptosis serves as a powerful selective pressure for the virus to evade. However, for the host, success is harsh and potentially costly, as apoptosis often contributes to pathogenesis. Here we examine some of the molecular mechanisms by which viruses manipulate the apoptotic machinery to their advantage and how we (as vertebrates) have evolved and learned to cope with viral evasion.
Similar content being viewed by others
Main
The apoptotic machinery
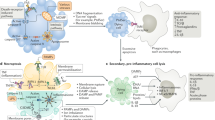
Apoptosis results from a collapse of cellular infrastructure through regulated internal proteolytic digestion, which leads to cytoskeletal disintegration, metabolic derangement and genomic fragmentation. Members of the cytosolic caspase family of proteinases (cysteine-based, aspartate-directed) form “the engine” of the apoptotic pathway1 (Fig. 1). The caspases (11 in total) represent one of more than 20 distinct components involved in initiation, execution and regulatory phases of the pathway, which indicates the extensive regulation this process has engendered over time. The apoptotic machinery shows sensitivity to a variety of agents by coordinating signals initiated by both internal sensors (intrinsic pathway, mitochondria-dependent) and external stimuli (extrinsic pathway, death receptor–mediated). When triggered, internal sensors (for example, p53) can initiate processes that result in the ultimate loss of mitochondrial integrity and apoptosis. Signals from the internal sensors are propagated to the mitochondria via pro-apoptotic Bcl-2 subfamily members (BH3 only), such as Bid, to oligomerize Bax and Bak, which are outer mitochondrial membrane proteins that promote the release of cytochrome c. Cytochrome c oligomerizes Apaf1 and recruits pro-caspase-9 (forming the apoptosome), which results in proteolytic conversion of pro-caspase-9 to an active enzyme. Caspase-9 then converts pro-caspase-3 to its active form, which, with other executioner caspases, such as caspase-7, then cleaves key substrates in the cell to orchestrate the cell's fatal collapse.
The intrinsic apoptotic pathway is initiated by internal sensors that monitor cellular stresses such as viral infection via activation of BH3 domain–only members of the Bcl-2 family. Activated BH3-only proteins are thought to mediate the assembly of pro-apoptotic members of the Bcl-2 family (Bax, Bak, Bcl-rambo, Bok) into hetero-oligomeric “pores” in the outer membrane of the mitochondria; this results in the release of factors such as cytochrome c, Smac (also known as Diablo) and Omi (also known as HtrA2) into the cytoplasm. The loss of mitochondrial membrane integrity can be blocked by the anti-apoptotic Bcl-2 family members Bcl-2, Bcl-xL, Bcl-w, Mcl-1 and Bcl-B. Release of cytochrome c promotes formation of the apoptosome, which contains Apaf-1 and pro-caspase-9. Autocatalytic activation of caspase-9 initiates the effector caspase cascade, which includes caspase-2, -3, -6 and/or -7. Caspase activation is negatively regulated by the IAPs, which are counter-balanced by the release of pro-apoptotic Smac and Omi from the mitochondria. The extrinsic pathway of apoptosis is triggered by TNF family death ligands binding to their cognate death receptors. Via their DDs, multimerized receptors interact with the DDs of adaptor proteins such as FADD. These adaptor proteins also contain DEDs that facilitate their binding to pro-caspase-8 and/or pro-caspase-10 to form the DISC. As part of the DISC, the pro-caspases are cleaved into their active forms and initiate the intrinsic pathway of apoptosis by cleaving Bid into t-Bid and activate the effector caspase cascade (caspase-3 is shown).
In contrast, the extrinsic pathway starts with members of the tumor necrosis factor (TNF) superfamily of death receptors transmitting external signals provided by immune effector cells to the virus-infected cell. Innate effector cells—for example, natural killer (NK) cells and dendritic cells (DCs)—can mount rapid antiviral responses through direct detection of viral products by pattern recognition receptors—such as binding of double-stranded RNA (dsRNA) by Toll-like receptor 3 (TLR3)—or by up-regulating death receptor ligands—for example, Fas ligand (FasL), TNF receptor–related apoptosis-inducing ligand (TRAIL) and TNF2. Later during adaptive immunity, these same death ligands are produced by antigen-specific cytotoxic T cells (CTLs).
The lethal program set in motion by these ligands mandates that their expression is transient and highly regulated at the transcriptional and post-translational steps. Upon binding TNF-family ligands, death receptors recruit adaptors and initiator caspases in a stepwise sequence based on specific interaction domains and form the death-inducing signaling complex (DISC) at the plasma membrane. In the case of Fas, the cytosolic adaptor FADD (Fas-associated death domain–containing protein) contains two interaction motifs: a death domain (DD) that associates with the homologous structure in Fas and a death effector domain (DED) that interacts with a homologous DED in pro-caspase-8 or pro-caspase-10. The proximal positioning of pro-caspase-8 and pro-caspase-10 in the DISC is thought to lead to autocatalysis and conversion to an active enzyme. Activated caspase-8 or caspase-10 can then directly convert pro-caspase-3 to its active form, completing the initiation phase of the pathway. There is cross-talk between the death receptor pathways and the mitochrondria-dependent arm through cleavage of the BH3-only protein Bid by caspase-8. In addition, the cytotoxic granule–associated proteinase granzyme B can bypass caspase-8 and, upon delivery by CTLs and NK effector cells, can directly cleave and activate caspase-3. Caspase-3 and other executioner caspases cleave numerous substrates, such as ICAD (inhibitor of caspase-activated DNase), which leads to genome fragmentation, collapse of the cell and preparation of cellular remnants for phagocytosis. Thus, vertebrates have evolved several distinct strategies to initiate apoptosis that lead to a common execution phase of the pathway.
All of the key pro-apoptotic components are preformed in the cell, enabling the pathway to respond rapidly to apoptosis-inducing signals. In contrast, the cellular regulatory elements of the death pathway often require new gene expression. The transcription factor NF-κB controls several genes, including those of the Bcl-2 and inhibitors of apoptosis (IAP) families, whose functions regulate apoptosis and promote cell survival3. This is especially pertinent when a virus has compromised the biosynthetic capacity of the host cell: death becomes the default outcome and, predictably, viral counter-strategies have evolved that mimic the key cellular regulatory elements. The key regulatory steps in the cell death pathway targeted by viral pathogens include control over the expression of internal sensors, cytochrome c release by Bcl-2 family members, inhibition of caspases and regulation of death receptor signaling. Indeed, the discovery of several of the key regulatory points in the apoptotic pathway have emerged directly from studies of the death-escape mechanisms used by viruses.
Blinding the sensors
The tumor suppressor p53 limits cellular proliferation by inducing apoptosis or cell cycle arrest in response to cellular stresses and is intimately linked to cancer development. p53-deficient mice are more prone to certain viral infections, indicating a role beyond tumor suppression4. Some of the genes whose transcription is stimulated by p53 include those that encode the death receptors Fas and TRAIL receptor 2 (TRAILR2), although the importance of their up-regulation by p53 in triggering apoptosis is unclear. Additionally, p53 induces the transcription of genes such as Bax, Bak1, Bbc3 and Pmaip1 whose products are involved in death signal propagation through the mitochondria5,6,7,8,9. p53 represses transcription of the anti-apoptotic protein Bcl-2, an antagonist of Bax and Bak10. Viruses can also disrupt apoptosis by inactivating p53. The SV40 large T antigen binds to p53 and sequesters it in an inactive complex11,12. The human papillomavirus E6 protein and adenovirus E1B-55K protein, in concert with E4orf6, promote ubiquitination and degradation of p53, albeit via different mechanisms13,14,15,16. Additionally, the pX protein encoded by hepatitis B virus complexes with p53 and inhibits p53-mediated transcriptional activation as well as p53-dependent apoptosis17.
Mimicking Bcl-2
Viral encoded orthologs of the anti-apoptotic regulator Bcl-2 are a widely used immune evasion strategy (Fig. 2). A pertinent example is adenovirus E1B-19K, which is similar both in sequence and function to Bcl-218,19. Unlike cell death signaled through cell-surface receptors, p53-mediated apoptosis in response to adenovirus infection does not require the cleavage of Bid into t-Bid to achieve Bax-Bak oligomerization and the subsequent release of cytochrome c. Instead, p53 stimulates a conformational change in Bax that is required for Bax-Bak interaction. These events are blocked by adenovirus E1B-19K as a result of its binding to Bak and the abrogation of Bax-Bak oligomerization20. E1B-19K also affects the apoptotic process signaled via TNF at the level of Bax and Bak activation. Treatment of cells with TNF produces a death signal that results in the cleavage of Bid to t-Bid followed by the recruitment of monomeric, pro-apoptotic Bax into a 500-kD protein complex and the release of cytochrome c from the mitochondria. In the case of TNF-mediated apoptosis, E1B-19K appears to interact primarily with Bax, inhibiting oligomerization and the subsequent release of cytochrome c21.
Cellular sensors, such as p53, that detect stress and initiate the apoptotic process are inactivated by the proteins adenovirus E1B-55K and human papillomavirus E6. Several viruses encode orthologs of Bcl-2 family proteins that antagonize their pro-apoptotic activity. Several viral strategies target the caspases. The vIAPs contain conserved BIR domains and their expression may shift the equilibrium of Smac to favor cell survival. Non-BIR–containing proteins, such as p35 and the serpins (including CrmA), also inhibit caspase activation.
Oncogenic human herpesviruses use Bcl-2 orthologs to block mitochondrial release of cytochrome c. This is also true for Epstein-Barr virus (EBV), which encodes two Bcl-2 orthologs (BHRF1 and BALF-122,23), and Kaposi's sarcoma–associated γ-herpesvirus (KSHV), which expresses KSbcl-224,25. Mouse γ-herpesvirus MHV-68, a virus that serves as a model for human EBV infection, encodes a Bcl-2 ortholog (MHVBcl-2) that protects against TNF-mediated apoptosis in cell culture. In addition, MHVBcl-2 is important for chronic infection, as demonstrated by the impaired virulence of MHV-68 virus lacking the Bcl-2 ortholog in interferon-γ (IFN-γ)–deficient mice26. In contrast, the human cytomegalovirus (CMV) UL37 gene product vMIA shares no sequence homology to Bcl-2; however, it resides in the mitochondria and appears to be functionally similar to Bcl-2, as it associates with the adenine nucleotide translocator and inhibits Fas-mediated apoptosis27.
Other viral proteins inhibit apoptosis by modulating Bcl-2 family members at the transcriptional level or via post-translational modification. The human T cell leukemia virus type 1 (HTLV-1) Tax protein transcriptionally activates the Bcl-xL promoter while repressing transcription of Bax28. HIV-1 Nef mediates phosphorylation of the pro-apoptotic Bad protein, abrogating its activity and suppressing apoptosis in T cells29. Similarly, the U(S)3 protein kinase encoded by herpes simplex virus 1 (HSV-1) mediates a post-translational modification of Bad and blocks its cleavage and subsequent activation of apoptosis30.
Caspase regulation
Caspases play a central role in apoptosis and are regulated in several ways31,32,33. The enzymatic activity of caspases is inhibited by a conserved family of inhibitor of apoptosis proteins (IAPs)34,35 that were originally defined in baculovirus based on the suppression of apoptosis and presence of a zinc-binding motif called a BIR (baculoviral IAP repeat)36. To date, eight cellular IAPs have been identified that regulate both the effector and initiator caspases37. For example, XIAP, c-IAP1, c-IAP2 and ML-IAP (livin) target the initiator caspase, caspase-9, and the effector caspases, caspase-3 and caspase-7. XIAP, c-IAP1 and c-IAP2 contain three BIR domains, each with a different function. The third BIR domain (BIR3) inhibits the activity of processed caspase-9, whereas the linker region between BIR1 and BIR2 abrogates the activity of caspase-3 and caspase-7. An IAP-ortholog strategy is used infrequently in mammalian viruses, with the exception of African swine fever virus, which encodes a viral IAP (vIAP) that does not contribute to virulence38. In mammals, the serpin CrmA—which is derived from cowpox and is present in most poxviruses—also inhibits several caspases, likely through covalent modification of caspase-8, and blocks or delays apoptosis in response to TNF and Fas signaling or CTLs39,40,41. Also, CrmA inhibits caspase-1, a critical processing enzyme for the inflammatory cytokine interleukin 1β (IL-1β).
Modulation of TNFR signaling
Several members of the TNF receptor (TNFR) superfamily, including Fas, TNFR1 and TRAILR2, are potent inducers of apoptosis42. Not surprisingly many viruses specifically target these cytokine receptors (Fig. 3). Neutralization of TNF by soluble decoy receptors was one of the first-described evasion tactics, as shown by the secreted TNFR2 ortholog expressed by Shope fibroma virus (rabbit poxvirus)43. Several TNFR orthologs have been identified in the genomes of lepri- and orthopoxviruses, including smallpox, which indicates its impact on the success of poxviruses44. The T2 protein of myxoma virus is a dimeric, high-affinity binding protein for TNF and virulence is attenuated when it is deleted from the viral genome45. Interestingly, the TNFR decoys in vaccinia, the attenuated form of smallpox, are mutated. Additionally, the poxviral TNFR ortholog T2 exists in an intracellular form that is required to inhibit apoptosis of lymphocytes46. Another TNFR ortholog is found in avian leukocytosis virus, which encodes an ortholog of TRAILR2 that serves as a virus entry factor47. Additionally, human CMV (but not mouse CMV) contains a TNFR ortholog encoded by the UL144 orf, although its functional significance remains obscure48.
Poxviruses block signaling via TNF and lymphotoxin by producing soluble decoy receptors (for example, myxoma virus T2 protein). vFLIPs contain two DEDs that interact with the homologous DED of FADD and pro-caspase-8 or pro-caspase-10, blocking caspase activation and inhibiting death receptor–induced apoptosis. The human CMV protein vICA binds and inhibits pro-caspase-8 activation, but lacks a DED. The EBV protein LMP1 self-aggregates and engages TRAF and TRADD molecules, activating “anti-apoptotic” NF-κB– and Jnk-dependent pathways. A heterocomplex of proteins encoded by the E3 region of adenovirus (E3-10.4K, E3-14.5K and E3-6.7K) facilitates the removal of the death receptors Fas, TRAILR1 and TRAILR2 from the surface of infected cells. Consequently, receptors accumulate in late endocytic compartments, and cells are desensitized to killing.
Herpes simplex virus 1, through its envelope glycoprotein D, uses a TNFR family member, herpesvirus entry mediator (HVEM), to gain access to the lymphoid compartment, where it can induce apoptosis of T cells49 and block maturation of antigen-presenting DCs50. The major B cell–transforming protein in EBV, LMP1, behaves like a constitutively activated CD40 by engaging TRAFs and TNFR1-associated DD protein (TRADD), which are adaptors used by TNFR to activate the transcription of anti-apoptotic genes through NF-κB and c-Jun NH2-terminal kinase (Jnk)-dependent signaling pathways. In a transgenic model, LMP1 expression prevents B cells from localizing to the follicle, thus protecting cells harboring latent virus from interactions with T cells51.
Whereas poxviruses inhibit TNF ligand-receptor interactions by deploying a soluble receptor smoke screen, a distinct strategy is used by adenoviruses. The E3 region encodes several proteins that sweep the cell surface clear of the death-inducing receptors Fas, TRAILR1 and TRAILR2. The E3 proteins responsible for modulating cell surface amounts of Fas are E3-10.4K and E3-14.5K52,53, which localize to various cellular membrane compartments, including the plasma membrane, as a heteromeric complex54. However, the ability of adenovirus to modulate TRAILR2 absolutely requires a third E3 protein, E3-6.7K, in addition to E3-10.4K and E3-14.5K55; this highlights the complexity of the mechanisms used by adenoviruses to inhibit signaling by these death receptors. Upon E3-induced down-regulation, death-inducing receptors accumulate in late endocytic compartments, resulting in the desensitization of infected cells to killing by FasL and TRAIL53,55.
Prevailing wisdom is that the E3-10.4K–E3-14.5K–E3-6.7K complex pirates the cellular endocytic compartments that direct membrane protein trafficking. A similar strategy is used by the HIV Nef protein, which down-modulates CD4 and major histocompatibility complex (MHC) class I from the cell surface by cross-linking these proteins to the cellular endocytic machinery56. As a consequence of Nef's actions, the membrane forms of TNF and the related cytokine LIGHT show sustained expression on the surface of T cells, potentially contributing to the cytopathic effects of HIV on the T cell compartment57. The substantial death of bystander (uninfected) lymphocytes during HIV infection may stimulate lymphopoiesis, possibly by homeostatic control mechanisms, which could provide a pool of dividing (but not HIV-specific) T cells for HIV genome replication58.
Viruses can down-modulate or enhance expression of death receptors and ligands to their own advantage. The loss of death receptors may function to protect infected cells from cytolysis by CTLs or NK cells, which express FasL or TRAIL upon activation59. In addition, this down-regulation may help inhibit apoptosis mediated by neighboring cells that are induced to express death receptor ligands upon infection. Induction of TRAIL has been observed in HCMV-50,60 and reovirus-infected61 cells and of FasL by HSV in T cells and HCMV in DCs49,50. It has even been proposed that this induction of FasL and TRAIL is another viral immune evasion tactic, through the killing of infiltrating host CTLs and DCs49,50. IFNs induce TRAIL in various cell types60,62,63; this suggests that a possible mechanism for up-regulation of TRAIL could be via production of IFNs by the virus-infected cell.
Several viruses have evolved a different strategy for blocking death receptor signaling at the level of DISC assembly; they do this through blockade of caspase-8 and caspase-10 processing. The viral FLICE (caspase-8) inhibitory proteins (FLIPs) contain DEDs, but lack caspase activity, and are present in the genomes of various γ-herpesviruses, including equine herpesvirus 2 (EHV-2), herpesvirus saimiri (HVS), KSHV, bovine herpesvirus 4 (BHV-4) and moluscum contagiosum virus (MCV)64,65. The cellular ortholog of vFLIP was subsequently cloned after identification of the vFLIPs65 and exists in both a ∼26-kD short (cFLIPS) and ∼55-kD long (cFLIPL) form generated by alternative splicing. cFLIPS is essentially the cellular ortholog of vFLIP and encodes two DEDs, whereas cFLIPL encodes an additional COOH-terminal domain with high homology to caspase-8 and caspase-10. Both isoforms of cFLIP are recruited to the DISC and inhibit death receptor–induced apoptosis; however, their mechanisms of action are slightly different: cFLIPS completely inhibits proteolytic processing of caspase-8 (similar to vFLIP) and cFLIPL allows partial caspase-8 processing66. The HCMV UL36 gene product vICA also associates with caspase-8 and blocks its activation, but shows no sequence identity to its proposed cellular orthologs, the FLIPs67.
vFLIPs interact with adaptor proteins that regulate the expression of NF-κB, including TNFR-associated factor 2 (TRAF2), receptor-interacting protein (RIP), NF-κB–inducing kinase (NIK) and the inhibitor of κB-kinase 2 (IKK2)68, which indicates that the vFLIPs may also regulate activation of transcription factors important in inhibiting apoptosis.
Death taxes
Cell death comes at a potentially large cost for the host. However, it is becoming appreciated that IFN-dependent nonapoptotic mechanisms can result in the successful attenuation of viral spread69. IFNs play a critical role in mounting innate responses to viral infection. IFNs are typically considered nonapoptotic but, in some circumstances, can promote apoptosis of virus-infected cells and are especially potent when they act in concert with TNF-related ligands. IFNs signal via the Janus protein kinase–signal transducers and activators of transcription pathway (Jak-STAT)70. Well studied IFN-inducible signaling pathways include the RNA-dependent protein kinase (PKR)71 and the 2′,5-oligoadenylate and RNase-L systems72. Upon interaction with virus-derived dsRNA, PKR is activated and can inhibit host-cell translation via phosphorylation of elongation initiation-factor 2α (eIF2α)73. More recently, it has been shown that PKR has additional activities, including the modulation of NF-κB74 and promotion of apoptosis by functional enhancement of the tumor suppressor genes p53 and IFN response factor 1 (IRF-1)75. The regulation of NF-κB by PKR places this protein at a critical crossroad in the coordination of apoptotic signals, potentially through the NF-κB–dependent induction of FasL and IFN-induced TRAIL.
Several noncytolytic antiviral programs activated by IFNs can arrest the viral life cycle at different steps, thereby attenuating virus spread and limiting the infection. However, IFNs and their response genes are themselves targeted by many viruses76. Replication-competent HCMV specifically inhibits induction of IFN-β transcription by an unknown mechanism77 that may involve inhibition of IRF-3 activation78. This blockade of IFN-β transcription is overridden by signaling via TNFR1 or the lymphotoxin β receptor (LTβR), but not by Fas or TRAIL receptors79. Induction of IFN-β by these two TRAF-adapting receptors is NF-κB–dependent, but occurs only in HCMV-infected cells. Therefore, host and virus factors cooperate to induce IFN-β, establishing a state of détente in which the host cell survives and the viral genome persists, but cannot produce new virions. This differs from the case of hepatitis B, where IFN signaling results in protease-dependent clearance of the viral genome69. These two examples highlight the virus- and cell type–specific issues often seen with IFN's antiviral action.
The diversity of strategies used by viruses to modulate the apoptotic pathway is as varied as there are viruses. In addition, for each virus, these strategies are integrated into a wider scheme that manipulates other aspects of host defenses. For example, herpesviruses have large DNA genomes that have accumulated an extensive repertoire of immune-evasion tactics that target both the afferent (for example, antigen recognition) and effector phases (apoptosis) of host defenses80. The cumulative effect of these mechanisms is thought to contribute to the ability of herpesviruses to sustain life-long infection. As a cautionary note, nearly all the data gathered to date are on clinically important viruses, which unfortunately often lack comparable animal models with which we can assess the role of immune-modulatory genes in viral pathogenesis. Indeed, there are significant differences in the molecular mechanisms used to modulate immune function by mouse CMV compared with those of human CMV. It is also important to recognize that a particular viral evasion strategy may act differently in a non-native host compared to in tissue culture, which also contributes to the difficulty in defining mechanisms of action. Nonetheless, the elucidation of evasion strategies directed at apoptotic mechanisms can provide deeper insight into the host-virus relationship that hopefully will yield better vaccine strategies.
References
Green, D.R. Apoptotic pathways: paper wraps stone blunts scissors. Cell 102, 1–4 (2000).
Medzhitov, R. Toll-like receptors and innate immunity. Nature Rev. Immunol. 1, 135–145 (2001).
Karin, M. & Lin, A. NF-κB at the crossroads of life and death. Nature Immunol. 3, 221–227 (2002).
Harvey, M. et al. Spontaneous and carcinogen-induced tumorigenesis in p53-deficient mice. Nature Genet. 5, 225–229 (1993).
Kannan, K. et al. DNA microarrays identification of primary and secondary target genes regulated by p53. Oncogene 20, 2225–2234 (2001).
Miyashita, T. & Reed, J.C. Tumor suppressor p53 is a direct transcriptional activator of the human bax gene. Cell 80, 293–299 (1995).
Nakano, K. & Vousden, K.H. PUMA, a novel proapoptotic gene, is induced by p53. Mol. Cell 7, 683–694 (2001).
Oda, E. et al. Noxa, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 288, 1053–1058 (2000).
Wu, G.S. et al. KILLER/DR5 is a DNA damage-inducible p53-regulated death receptor gene. Nature Genet. 17, 141–143 (1997).
Gross, A., McDonnell, J.M. & Korsmeyer, S.J. BCL-2 family members and the mitochondria in apoptosis. Genes Dev. 13, 1899–1911 (1999).
Lane, D.P. & Crawford, L.V. T antigen is bound to a host protein in SV40-transformed cells. Nature 278, 261–263 (1979).
Linzer, D.I. & Levine, A.J. Characterization of a 54K dalton cellular SV40 tumor antigen present in SV40-transformed cells and uninfected embryonal carcinoma cells. Cell 17, 43–52 (1979).
Steegenga, W.T., Riteco, N., Jochemsen, A.G., Fallaux, F.J. & Bos, J.L. The large E1B protein together with the E4orf6 protein target p53 for active degradation in adenovirus infected cells. Oncogene 16, 349–357 (1998).
Querido, E. et al. Degradation of p53 by adenovirus E4orf6 and E1B55K proteins occurs via a novel mechanism involving a Cullin-containing complex. Genes Dev. 15, 3104–3117 (2001).
Scheffner, M., Werness, B.A., Huibregtse, J.M., Levine, A.J. & Howley, P.M. The E6 oncoprotein encoded by human papillomavirus types 16 and 18 promotes the degradation of p53. Cell 63, 1129–1136 (1990).
Werness, B.A., Levine, A.J. & Howley, P.M. Association of human papillomavirus types 16 and 18 E6 proteins with p53. Science 248, 76–79 (1990).
Wang, X.W. et al. Abrogation of p53-induced apoptosis by the hepatitis B virus X gene. Cancer Res. 55, 6012–6016 (1995).
White, E., Cipriani, R., Sabbatini, P. & Denton, A. Adenovirus E1B 19-kilodalton protein overcomes the cytotoxicity of E1A proteins. J. Virol. 65, 2968–2978 (1991).
White, E. Regulation of apoptosis by adenovirus E1A and E1B oncogenes. Semin. Virol. 8, 505–513 (1998).
Henry, H., Thomas, A., Shen, Y. & White, E. Regulation of the mitochondrial checkpoint in p53-mediated apoptosis confers resistance to cell death. Oncogene 21, 748–760 (2002).
Sundararajan, R. & White, E. E1B 19K blocks Bax oligomerization and tumor necrosis factor α- mediated apoptosis. J. Virol. 75, 7506–7516 (2001).
Henderson, S. et al. Epstein-Barr virus-coded BHRF1 protein, a viral homologue of Bcl-2, protects human B cells from programmed cell death. Proc. Natl. Acad. Sci. USA 90, 8479–8483 (1993).
Marshall, W.L., Datta, R., Hanify, K., Teng, E. & Finberg, R.W. U937 cells overexpressing bcl-xL are resistant to human immunodeficiency virus-1-induced apoptosis and human immunodeficiency virus-1 replication. Virology 256, 1–7 (1999).
Sarid, R., Sato, T., Bohenzky, R.A., Russo, J.J. & Chang, Y. Kaposi's sarcoma-associated herpesvirus encodes a functional bcl-2 homologue. Nature Med. 3, 293–298 (1997).
Cheng, E.H. et al. A Bcl-2 homolog encoded by Kaposi sarcoma-associated virus, human herpesvirus 8, inhibits apoptosis but does not heterodimerize with Bax or Bak. Proc. Natl. Acad. Sci. USA 94, 690–694 (1997).
Gangappa, S., van Dyk, L.F., Jewett, T.J., Speck, S.H. & Virgin, H.W. Identification of the in vivo role of a viral bcl-2. J. Exp. Med. 195, 931–940 (2002).
Goldmacher, V.S. et al. A cytomegalovirus-encoded mitochondria-localized inhibitor of apoptosis structurally unrelated to Bcl-2. Proc. Natl. Acad. Sci. USA 96, 12536–12541 (1999).
Tsukahara, T. et al. Induction of Bcl-xL expression by human T-cell leukemia virus type 1 Tax through NF-κB in apoptosis-resistant T-cell transfectants with Tax. J. Virol. 73, 7981–7987 (1999).
Wolf, D. et al. HIV-1 Nef associated PAK and PI3-kinases stimulate Akt-independent Bad- phosphorylation to induce anti-apoptotic signals. Nature Med. 7, 1217–1224 (2001).
Munger, J. & Roizman, B. The US3 protein kinase of herpes simplex virus 1 mediates the posttranslational modification of BAD and prevents BAD-induced programmed cell death in the absence of other viral proteins. Proc. Natl. Acad. Sci. USA 98, 10410–10415 (2001).
Earnshaw, W.C., Martins, L.M. & Kaufmann, S.H. Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu. Rev. Biochem. 68, 383–424 (1999).
Huang, H. et al. The inhibitor of apoptosis, cIAP2, functions as a ubiquitin-protein ligase and promotes in vitro monoubiquitination of caspases 3 and 7. J. Biol. Chem. 275, 26661–26664 (2000).
Suzuki, Y., Nakabayashi, Y. & Takahashi, R. Ubiquitin-protein ligase activity of X-linked inhibitor of apoptosis protein promotes proteasomal degradation of caspase-3 and enhances its anti-apoptotic effect in Fas-induced cell death. Proc. Natl. Acad. Sci. USA 98, 8662–8667 (2001).
Deveraux, Q.L. & Reed, J.C. IAP family proteins–suppressors of apoptosis. Genes Dev. 13, 239–252 (1999).
Hay, B.A. Understanding IAP function and regulation: a view from Drosophila. Cell Death Differ. 7, 1045–1056 (2000).
Clem, R.J. Baculoviruses and apoptosis: the good, the bad, and the ugly. Cell Death Differ. 8, 137–143 (2001).
Shi, Y. Mechanisms of caspase activation and inhibition during apoptosis. Mol. Cell 9, 459–470 (2002).
Neilan, J.G. et al. An African swine fever virus gene with similarity to the proto-oncogene bcl-2 and the Epstein-Barr virus gene BHRF1. J. Virol. 67, 4391–4394 (1993).
Miura, M., Friedlander, R.M. & Yuan, J. Tumor necrosis factor-induced apoptosis is mediated by a CrmA-sensitive cell death pathway. Proc. Natl. Acad. Sci. USA 92, 8318–8322 (1995).
Talley, A.K. et al. Tumor necrosis factor α-induced apoptosis in human neuronal cells: protection by the antioxidant N-acetylcysteine and the genes bcl-2 and crmA. Mol. Cell. Biol. 15, 2359–2366 (1995).
Tewari, M. & Dixit, V.M. Fas- and tumor necrosis factor-induced apoptosis is inhibited by the poxvirus crmA gene product. J. Biol. Chem. 270, 3255–3260 (1995).
Wallach, D. et al. Tumor necrosis factor receptor and Fas signaling mechanisms. Annu. Rev. Immunol. 17, 331–367 (1999).
Smith, C.A. et al. A receptor for tumor necrosis factor defines an unusual family of cellular and viral proteins. Science 248, 1019–1024 (1990).
Reading, P.C., Khanna, A. & Smith, G.L. Vaccinia virus CrmE encodes a soluble and cell surface tumor necrosis factor receptor that contributes to virus virulence. Virology 292, 285–298 (2002).
Upton, C., Macen, J., Schreiber, M. & McFadden, G. Myxoma virus expresses a secreted protein with homology to the tumor necrosis factor receptor gene family that contributes to viral virulence. Virology 184, 370–382 (1991).
Schreiber, M., Sedger, L. & McFadden, G. Distinct domains of M-T2, the myxoma virus tumor necrosis factor (TNF) receptor homolog, mediate extracellular TNF binding and intracellular apoptosis inhibition. J. Virol. 71, 2171–2181 (1997).
Brojatsch, J., Naughton, J., Rolls, M.M., Zingler, K. & Young, J.A. CAR1, a TNFR-related protein, is a cellular receptor for cytopathic avian leukosis-sarcoma viruses and mediates apoptosis. Cell 87, 845–855 (1996).
Benedict, C. et al. Cutting edge: A novel viral TNF receptor superfamily member in virulent strains of human cytomegalovirus. J. Immunol. 126, 6967–6970 (1999).
Raftery, M.J. et al. Herpes simplex virus type 1 infection of activated cytotoxic T cells: Induction of fratricide as a mechanism of viral immune evasion. J. Exp. Med. 190, 1103–1104 (1999).
Raftery, M.J. et al. Targeting the function of mature dendritic cells by human cytomegalovirus: a multilayered viral defense strategy. Immunity 15, 997–1009 (2001).
Uchida, J. et al. Mimicry of CD40 signals by Epstein-Barr virus LMP1 in B lymphocyte responses. Science 286, 300–303 (1999).
Shisler, J., Yang, C., Walter, B., Ware, C. & Gooding, L. The adenovirus E3-10. 4K/14. 5K complex mediates loss of cell surface fas (CD95) and resistance to fas-induced apoptosis. J. Virol. 71, 8299–8306 (1997).
Tollefson, A.E. et al. Forced degradation of Fas inhibits apoptosis in adenovirus-infected cells. Nature 392, 726–730 (1998).
Stewart, A.R., Tollefson, A.E., Krajcsi, P., Yei, S.P. & Wold, W.S. The adenovirus E3 10.4K and 14.5K proteins, which function to prevent cytolysis by tumor necrosis factor and to down-regulate the epidermal growth factor receptor, are localized in the plasma membrane. J. Virol. 69, 172–181 (1995).
Benedict, C.A. et al. Three adenovirus E3 proteins cooperate to evade apoptosis by tumor necrosis factor-related apoptosis-inducing ligand receptor-1 and -2. J. Biol. Chem. 276, 3270–3278 (2001).
Piguet, V., Schwartz, O., Le Gall, S. & Trono, D. The downregulation of CD4 and MHC-I by primate lentiviruses: a paradigm for the modulation of cell surface receptors. Immunol. Rev. 168, 51–63 (1999).
Lama, J. & Ware, C.F. Human immunodeficiency virus type 1 Nef mediates sustained membrane expression of tumor necrosis factor and the related cytokine LIGHT on activated T cells. J. Virol. 74, 9396–9402 (2000).
Overbaugh, J. & Bangham, C.R.M. Selection forces and constraints on retroviral sequence variation. Science 292, 1106–1109 (2001).
Ashkenazi, A. & Dixit, V.M. Apoptosis control by death and decoy receptors. Curr. Opin. Cell. Biol. 11, 255–260 (1999).
Sedger, L.M. et al. IFN-γ mediates a novel antiviral activity through dynamic modulation of TRAIL and TRAIL receptor expression. J. Immunol. 163, 920–926 (1999).
Vidalain, P.O. et al. Measles virus induces functional TRAIL production by human dendritic cells. J. Virol. 74, 556–559 (2000).
Kayagaki, N. et al. Expression and function of TNF-related apoptosis-inducing ligand on murine activated NK cells. J. Immunol. 163, 1906–1913 (1999).
Fanger, N.A., Maliszewski, C.R., Schooley, K. & Griffith, T.S. Human dendritic cells mediate cellular apoptosis via tumor necrosis factor-related apoptosis-inducing ligand (TRAIL). J. Exp. Med. 190, 1155–1164 (1999).
Bertin, J. et al. Death effector domain-containing herpesvirus and poxvirus proteins inhibit both Fas- and TNFR1-induced apoptosis. Proc. Natl. Acad. Sci. USA 94, 1172–1176 (1997).
Thome, M. & Tschopp, J. Regulation of lymphocyte proliferation and death by FLIP. Nature Rev. Immunol. 1, 50–58 (2001).
Krueger, A., Schmitz, I., Baumann, S., Krammer, P.H. & Kirchhoff, S. Cellular FLICE-inhibitory protein splice variants inhibit different steps of caspase-8 activation at the CD95 death-inducing signaling complex. J. Biol. Chem. 276, 20633–20640 (2001).
Skaletskaya, A. et al. A cytomegalovirus-encoded inhibitor of apoptosis that suppresses caspase-8 activation. Proc. Natl. Acad. Sci. USA 7829–7834 (2001).
Chaudhary, P.M., Jasmin, A., Eby, M.T. & Hood, L. Modulation of the NF-κB pathway by virally encoded death effector domains-containing proteins. Oncogene 18, 5738–5746 (1999).
Guidotti, L.G. & Chisari, F.V. Noncytolytic control of viral infections by the innate and adaptive immune response. Annu. Rev. Immunol. 19, 65–91 (2001).
Darnell, J.E. Jr. Studies of IFN-induced transcriptional activation uncover the Jak-Stat pathway. J. Interferon Cytokine Res. 18, 549–554 (1998).
Tan, S.L. & Katze, M.G. The emerging role of the interferon-induced PKR protein kinase as an apoptotic effector: a new face of death? J. Interferon Cytokine Res. 19, 543–554 (1999).
Castelli, J.C. et al. A study of the interferon antiviral mechanism: apoptosis activation by the 2–5A system. J. Exp. Med. 186, 967–972 (1997).
Meurs, E. et al. Molecular cloning and characterization of the human double-stranded RNA-activated protein kinase induced by interferon. Cell 62, 379–390 (1990).
Chu, W.M. et al. JNK2 and IKKβ are required for activating the innate response to viral infection. Immunity 11, 721–731 (1999).
Jagus, R., Joshi, B. & Barber, G.N. PKR, apoptosis and cancer. Int. J. Biochem. Cell. Biol. 31, 123–138 (1999).
Barber, G.N. Host defense, viruses and apoptosis. Cell Death Differ. 8, 113–126 (2001).
Browne, E.P., Wing, B., Coleman, D. & Shenk, T. Altered cellular mRNA levels in human cytomegalovirus-infected fibroblasts: viral block to the accumulation of antiviral mRNAs. J. Virol. 75, 12319–12330 (2001).
Preston, C.M., Harman, A.N. & Nicholl, M.J. Activation of interferon response factor-3 in human cells infected with herpes simplex virus type 1 or human cytomegalovirus. J. Virol. 75, 8909–8916 (2001).
Benedict, C.A. et al. Lymphotoxins and cytomegalovirus cooperatively induce interferon-β, establishing host-virus détente. Immunity 15, 617–626 (2001).
Tortorella, D., Gewurz, B.E., Furman, M.H., Schust, D.J. & Ploegh, H.L. Viral subversion of the immune system. Annu. Rev. Immunol. 18, 861–926 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Benedict, C., Norris, P. & Ware, C. To kill or be killed: viral evasion of apoptosis. Nat Immunol 3, 1013–1018 (2002). https://doi.org/10.1038/ni1102-1013
Issue Date:
DOI: https://doi.org/10.1038/ni1102-1013
This article is cited by
-
Prediction of motif-mediated viral mimicry through the integration of host–pathogen interactions
Archives of Microbiology (2024)
-
Duck Tembusu virus infection induces mitochondrial-mediated and death receptor-mediated apoptosis in duck embryo fibroblasts
Veterinary Research (2022)
-
Apoptotic caspase inhibits innate immune signaling by cleaving NF-κBs in both Mammals and Flies
Cell Death & Disease (2022)
-
A long way to go: caspase inhibitors in clinical use
Cell Death & Disease (2021)
-
Stress proteins: the biological functions in virus infection, present and challenges for target-based antiviral drug development
Signal Transduction and Targeted Therapy (2020)