Abstract
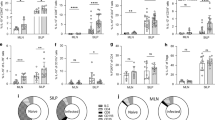
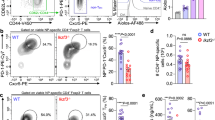
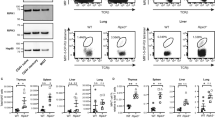
MURINE T-helper clones are classified into two distinct subsets (Thl and Th2) on the basis of their patterns of lymphokine secretion. Thl clones secrete interleukin-2 (IL-2), tumour necrosis factor-β (TNF-β) and interferon-γ (IFN-γ), whereas Th2 clones secrete IL-4, IL-5 and IL-10 (ref. 1). These subsets are reciprocally regulated by IL-4, IL-10 and IFN-γ and differentially promote antibody or delayed-type hypersensitivity responses2,3. To evaluate whether IL-4 is required for mounting Th2 responses, we generated IL-4-mutant mice (IL-4–/ –)4,5 and assessed the cytokine secretion pattern of T cells both from naive and Nippostrongylus brasiliensis infected mice. CD4+ T cells from naive IL-4–/ – mice failed to produce Th2-derived cytokines after in vitro stimulation. The levels of Th2 cytokines IL-5, IL-9 and IL-10 from CD4+ T cells obtained after nematode infection were significantly reduced. The reduced IL-5 production in IL-4–/ –mice correlated with reduced helminth-induced eosinophilia, which has been shown to be dependent on IL-5 in vivo6. We conclude that IL-4 is required for the generation of the Th2-derived cytokines and that immune responses dependent on these cytokines are impaired.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mosmann, T. R., Cherwinski, H., Bond, M. W., Giedlin, M. A. & Coffman, R. L. J. Immun. 136, 2348–2357 (1986).
Mosmann, T. R. & Coffman, R. L. A. Rev. Immun. 7, 145–173 (1989).
Fiorentino, D. F., Bond, M. W. & Mosmann, T. R. J. exp. Med. 170, 2081–2095 (1989).
Thomas, K. R. & Capecchi, M. R. Cell 51, 503–512 (1987).
Doetschmann, T. et al. Nature 330, 376–578 (1987).
Coffman, R. L., Seymour, B. W. P., Hudak, S., Jackson, J. & Rennick, D. Science 245, 308–310 (1989).
Lichtman, A. H., Kurt-Jones, E. A. & Abbas, A. K. Proc. natn. Acad. Sci. U.S.A. 84, 824–827 (1987).
Le-Gros, G., Ben-Sasson, S. Z., Seder, R., Finkelman, F. D. & Paul, W. E. J. exp. Med. 172, 921–929 (1990).
Swain, S. L., Weinberg, A. D., English, B. & Huston, G. J. Immun. 3796–3806 (1990).
Street, N. E. et al. J. Immun. 144, 1629–1639 (1990).
Svetic, A. et al. J. Immun. 147, 2391–2397 (1991).
Gajewski, T. F. & Fitch, F. W. J. Immun. 140, 4245–4252 (1988).
Moskophidis, D. & Lehmann-Grube, F. Proc. natn. Acad. Sci. U.S.A. 86, 3291–3295 (1989).
Fung-Leung, W.-P., Kündig, T. M., Zinkernagel, R. M. & Mak, T. W. J. exp. Med. 174, 1425–1429 (1991).
Finkelman, F. D. et al. A. Rev. Immun. 8, 303–333 (1990).
Finkelman, F. D. et al. J. Immun. 141, 2335–2341 (1988).
Kühn, R., Rajewsky, K. & Müller, W. Science 254, 707–710 (1991).
Finkelman, F. D. et al. Int. Immun. 3, 599–607 (1991).
DeKruyff, R. H., Mosmann, T. R. & Umetsu, D. T. Eur. J. Immun. 20, 2219–2227 (1990).
Plaut, M. et al. Nature 339, 64–67 (1989).
Mansour, S. L., Thomas, K. R. & Capecchi, M. R. Nature 336, 348–352 (1988).
Doetschmann, T., Eistetter, M., Katz, M., Schmidt, W. & Kemler, R. J. J. Embryol. exp. Morph. 87, 27–45 (1985).
Seder, R. A., Le-Gros, G., Ben-Sasson, S. A., Urban, J. J. & Finkelman, F. D. Eur. J. Immun. 21, 1241–1247 (1991).
Sarmiento, M., Glasebrook, A. L. & Fitch, F. W. J. Immun. 125, 2665–2672 (1980).
Dialynas, D. P. et al. J. Immun. 131, 2445–2451 (1983).
Leo, O., Foo, M., Sachs, D., Samelson, L. E. & Bluestone, J. A. Proc. natn. Acad. Sci. U.S.A. 84, 1374–1378 (1987).
Ohara, J. & Paul, W. E. Nature 315, 333–336 (1985).
Schmitt, E., van Brandwijk, R., Van Snick, J., Siebold, B. & Ruede, E. Eur. J. Immun. 19, 2167–2170 (1989).
Jennings, F. W., Mulligan, W. & Urquhart, G. M. Expl Parasitol. 13, 367–371 (1963).
Baniyash, M. & Eshhar, Z. Eur. J. Immun. 14, 799–807 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kopf, M., Gros, G., Bachmann, M. et al. Disruption of the murine IL-4 gene blocks Th2 cytokine responses. Nature 362, 245–248 (1993). https://doi.org/10.1038/362245a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/362245a0
This article is cited by
-
The brain cytokine orchestra in multiple sclerosis: from neuroinflammation to synaptopathology
Molecular Brain (2024)
-
Current and Emerging Strategies to Inhibit Type 2 Inflammation in Atopic Dermatitis
Dermatology and Therapy (2022)
-
Indicating biologics for chronic rhinosinusitis with nasal polyps (CRSwNP)
Allergo Journal International (2022)
-
Dokumentation von Biologika-Therapien bei chronischer Rhinosinusitis mit Polyposis nasi (CRSwNP): Dupilumab, Omalizumab und Mepolizumab
Allergo Journal (2022)
-
Interleukin-31 promotes fibrosis and T helper 2 polarization in systemic sclerosis
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.