Abstract
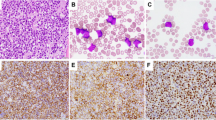
To define reproducible criteria for subgroups of diffuse large B-cell lymphomas (DLBCL), including lymphomas with plasmablastic/plasmacytoid features (PB/PC-Fs), we investigated 66 DLBCL; the samples were categorized as either centroblastic (CB), immunoblastic (IB) or PB/PC-F applying standardized morphologic criteria. Blinded specimens were reviewed by three independent pathologists. The final consensus classification included 44 CB (67%), seven IB (10%) and 15 PB/PC-F (23%). The interobserver agreement between two centers (Vienna, Würzburg) was 93.5%. Most PB/PC-F were CD20+, cIgM+, MUM-1+, CD138±, bcl-6−, corresponding to an activated B-cell phenotype. Immunoglobulin-VH gene mutation analysis was consistent with a germinal or postgerminal center-cell origin. By fluorescence in situ hybridization analysis, 11/13 (85%) PB/PC-F had a monoallelic TP53 deletion. The pretreatment characteristics of patients with PB/PC-F included a tendency for more B symptoms, extranodal disease and a higher IPI. Importantly, PB/PC-F were resistant to standard chemotherapy (complete remission rate 47%, relapse rate 71%) and even autologous stem-cell transplantation. The median overall survival (OS) (14 months, P<0.002) and disease-free survival (6 months, P=0.02) were significantly shorter compared to patients with CB and IB. The OS difference was pronounced within the low and low-intermediate IPI risk group (P<0.001). Our data indicate a strong association of plasmablastic/plasmacytoid morphology with TP53 deletions, poor response to chemotherapy and short survival.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Harris NL, Jaffe ES, Stein H, Banks PM, Chan JK, Cleary ML et al. A revised European–American classification of lymphoid neoplasms: a proposal from the international lymphoma study group. Blood 1994; 84: 1361–1392.
Harris NL, Jaffe ES, Diebold J, Flandrin G, Muller-Hermelink HK, Vardiman J et al. World Health Organization classification of neoplastic diseases of the hematopoietic and lymphoid tissues: report of the clinical advisory committee meeting – Airle House, Virginia, November 1997. J Clin Oncol 1999; 17: 3835–3849.
Gatter KC, Warnke RA . Diffuse large B-cell lymphoma. In: Jaffe ES, Harris NL, Stein H, Vardiman JW (eds). World Health Organization Classification of Tumours, Pathology and Genetics of Tumours of the Haematopoietic and Lymphoid Tissues. Lyon, France: IARC Press, 2001, pp 171–174.
Fisher RI, Gaynor ER, Dahlberg S, Oken MM, Grogan TM, Mize EM et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non Hodgkin's lymphoma. N Engl J Med 1993; 328: 987–1006.
Miller TP, Dahlberg S, Cassady JR, Adelstein DJ, Spier CM, Grogan TM et al. Chemotherapy alone compared with chemotherapy plus radiotherapy for localized intermediate- and high-grade non Hodgkin's lymphoma. N Engl J Med 1998; 239: 21–26.
Armitage JO, Weisenburger DD . New approach to classifying non Hodgkin's lymphomas: clinical features of the major histologic subtypes. Non-Hodgkin's lymphoma classification project. J Clin Oncol 1998; 16: 2780–2795.
Lennert K, Feller AC . Histopathology of non Hodgkin's lymphomas (based on the updated Kiel classification) with a section of clinical therapy. In: M Engelhard, G Brittinger (eds). Berlin: Springer-Verlag, 1992, 115–127.
Engelhard M, Brittinger G, Huhn D, Gerhartz HH, Meusers P, Siegert W et al. Subclassification of diffuse large B-cell lymphomas according to the Kiel classification: distinction of centroblastic and immunoblastic lymphomas is a significant prognostic risk factor. Blood 1997; 89: 2291–2297.
Schlegelberger B, Zwingers T, Harder L, Nowotny H, Siebert R, Vesely M et al. Clinicopathogenetic significance of chromosomal abnormalities in patients with blastic peripheral B-cell lymphoma. Blood 1999; 94: 3114–3120.
Alizadeh AA, Eisen MB, Davis RE, Ma C, Lossos IS, Rosenwald A et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 2000; 403: 503–511.
Rosenwald A, Wright G, Chan WC, Connors J, Campo E, Fisher RI et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med 2002; 346: 1937–1947.
Colomo L, López-Guillermo A, Perales M, Rives S, Martinez A, Bosch F et al. Clinical impact of the differentiation profile assessed by immunophenotyping in patients with diffuse large B-cell lymphoma. Blood 2003; 101: 78–84.
Delecluse HJ, Anagnostopoulos I, Dallenbach F, Hummel M, Marafioti T, Schneider U et al. Plasmablastic lymphomas of the oral cavity: a new entity associated with the human immunodeficiency virus infection. Blood 1997; 89: 1413–1420.
Dupin N, Diss TL, Kellam P, Tulliez M, Du MQ, Sicard D et al. HHV-8 is associated with a plasmablastic variant of Castleman disease that is linked to HHV-8-positive plasmablastic lymphoma. Blood 2000; 95: 1406–1412.
De Paepe P, Baens M, Van Krieken H, Verhasselt B, Stul M, Simons A et al. ALK activation by the CTLC-ALK fusion is a recurrent event in large B-cell lymphoma. Blood 2003; 102: 2638–2641.
Gascoyne RD, Lamant L, Martin-Subero JI, Valia S, Lestou VS, Harris NL et al. ALK-positive diffuse large B-cell lymphoma is associated with Clathrin-ALK rearrangements: report of six cases. Blood 2003; 102: 2568–2573.
Onciu M, Behm FG, Downing JR, Shurtleff SA, Raimondi SC, Ma Z, Morris SW et al. ALK-positive plasmablastic B-cell lymphoma with expression of the NPM-ALK fusion transcript: report of two cases. Blood 2003; 102: 2642–2644.
Bonadonna G, Monfardini S . Chemotherapy on non-Hodgkin's lymphomas. Cancer Treat Rev 1974; 1: 167–181.
Longo DL, DeVita Jr VT, Duffey PL, Wesley MN, Ihde DC, Hubbard SM et al. Superiority of ProMACE-CytaBOM over ProMACE-MOPP in the treatment of advanced diffuse aggressive lymphoma: results of a prospective randomized trial. J Clin Oncol 1991; 9: 25–38.
Cabanillas F, Hagemeister FB, Bodey GP, Freireich EJ . IMVP-16: an effective regimen for patients with lymphoma who have relapsed after initial combination chemotherapy. Blood 1982; 60: 693–697.
Fridrik MA, Hausmaninger H, Linkesch W, Stoger M, Sill H, Neubauer M et al. CEOP-IMVP-Dexa in the treatment of aggressive lymphomas: an Austrian multicenter trial. J Clin Oncol 1996; 14: 227–232.
Cheson BD, Horning SJ, Coiffier B, Shipp MA, Fisher RI, Connors JM et al. Report of an international workshop to standardize response criteria for non-Hodgkin's lymphomas. J Clin Oncol 1999; 17: 1244.
Velasquez WS, Cabanillas F, Salvador P, McLaughlin P, Fridrik M, Tucker S et al. Effective salvage therapy for lymphoma with cisplatin in combination with high dose Ara-C and dexamethasone (DHAP). Blood 1988; 71: 117–122.
Colwill R, Crump M, Couture F, Danish R, Stewart AK, Sutton DM et al. Mini-BEAM as salvage therapy for relapsed or refractory Hodgkin's disease before intensive therapy and autologous bone marrow transplantation. J Clin Oncol 1995; 13: 396–402.
Apperley JF, Girinsky T, Friedrich Wl . Conditioning regimens. In: Apperley JF, Gluckman E, Gratwohl A (eds). Blood and Marrow Transplantation. Paris: ESH, 1998, pp 98–115.
Kaplan EL, Meier P . Non parametric estimation from incomplete observation. J Am Stat Assoc 1958; 53: 457–481.
Cox DR . Regression models and life-tables. J R Stat Soc 1972; 34: 187–220.
Chott A, Haedicke W, Mosberger I, Fodinger M, Winkler K, Mannhalter C et al. Most CD56+ intestinal lymphomas are CD8+CD5− T-cell lymphomas of monomorphic small to medium size histology. Am J Pathol 1998; 153: 1483–1490.
Drach J, Ackermann J, Fritz E, Kromer E, Schuster R, Gisslinger H et al. Presence of a p53 gene deletion in patients with multiple myeloma predicts for short survival after conventional-dose chemotherapy. Blood 1998; 92: 802–809.
Welzel N, Le T, Marculescu R, Mitterbauer G, Chott A, Pott C et al. Templated nucleotide addition and immunoglobulin JH-gene utilization in t(11;14) junctions: implications for the mechanism of translocation and the origin of mantle cell lymphoma. Cancer Res 2001; 61: 1629–1636.
Falini B, Fizzotti M, Pucciarini A, Bigerna B, Marafioti T, Gambacorta M et al. A monoclonal antibody (MUM1p) detects expression of the MUM1/IRF4 protein in a subset of germinal center B cells, plasma cells, and activated T cells. Blood 2000; 95: 2084–2092.
Lossos IS, Jones CD, Warnke R, Natkunam Y, Kaizer H, Zehnder JL et al. Expression of a single gene, BCL-6, strongly predicts survival in patients with diffuse large B-cell lymphoma. Blood 2001; 98: 945–951.
Hsu FJ, Levy R . Preferential use of the VH4 Ig gene family by diffuse large-cell lymphoma. Blood 1995; 86: 3072–3082.
Lossos IS, Okada CY, Tibshirani R, Warnke R, Vose JM, Greiner TC et al. Molecular analysis of immunoglobulin genes in diffuse large B-cell lymphomas. Blood 2000; 95: 1797–1803.
Ichikawa A, Kinoshita T, Watanabe T, Kato H, Nagai H, Tsushita K et al. Mutations of the p53 gene as a prognostic factor in aggressive B-cell lymphoma. N Engl J Med 1997; 337: 529–534.
Offit K, Lo Coco F, Louie DC, Parsa NZ, Leung D, Portlock C et al. Rearrangement of the bcl-6 gene as a prognostic marker in diffuse large-cell lymphoma. N Engl J Med 1994; 331: 74–80.
Filipits M, Stranzl T, Pohl G, Heinzl H, Jager U, Geissler K et al. Drug resistance factors in acute myeloid leukemia: a comparative analysis. Leukemia 2000; 14: 68–76.
Coiffier B, Lepage E, Brière J, Herbrecht R, Tilly H, Bouabdallah R et al CHOP chemotherapy plus Rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med 2002; 346: 235–242.
Greipp PR, Leong T, Bennett JM, Gaillard JP, Klein B, Stewart JA et al. Plasmablastic morphology – an independent prognostic factor with clinical and laboratory correlates: Eastern Cooperative Oncology Group (ECOG) myeloma trial E9486 report by the ECOG Myeloma Laboratory Group. Blood 1998; 91: 2501–2507.
Lamant L, Pulford K, Bischof D, Morris SW, Mason DY, Delsol G et al. Expression of the ALK tyrosine kinase gene in neuroblastoma. Am J Pathol 2000; 156: 1711–1721.
Acknowledgements
IS-K and IH contributed equally to this work and should both be considered first authors. This work was supported by a grant from the ‘ICP Program of the Austrian Federal Ministry for Education, Science and Culture, by Grant P13984-GEN from the ‘Fonds zur Förderung der wissenschaftlichen Forschung’ and by a grant from the Kommission Onkologie of the Medical Faculty of the University of Vienna. Expert technical assistance by Susanne Hagmann, Ulrike Zeman and Isabella Mosberger is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Simonitsch-Klupp, I., Hauser, I., Ott, G. et al. Diffuse large B-cell lymphomas with plasmablastic/plasmacytoid features are associated with TP53 deletions and poor clinical outcome. Leukemia 18, 146–155 (2004). https://doi.org/10.1038/sj.leu.2403206
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403206
Keywords
This article is cited by
-
Lymphomas with plasmablastic features: a report of the lymphoma workshop of the 20th meeting of the European Association for Haematopathology
Virchows Archiv (2023)
-
Plasmablastic myeloma in Taiwan frequently presents with extramedullary and extranodal mass mimicking plasmablastic lymphoma
Virchows Archiv (2022)
-
MYC: a multipurpose oncogene with prognostic and therapeutic implications in blood malignancies
Journal of Hematology & Oncology (2021)
-
Clinical presentation and characteristics of lymphoma in the head and neck region
Head & Face Medicine (2019)
-
Clinical aspects of TP53 gene inactivation in diffuse large B-cell lymphoma
BMC Medical Genomics (2019)