Abstract
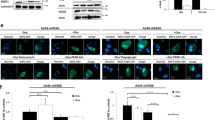
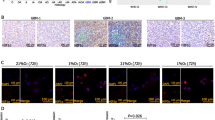
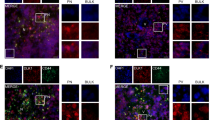
Hypoxic regions within solid tumors are often resistant to chemotherapy and radiation. BNIP3 (Bcl-2/E1B 19 kDa interacting protein) is a proapoptotic member of the Bcl-2 family that is expressed in hypoxic regions of tumors. During hypoxia, BNIP3 expression is increased in many cell types and upon forced overexpression BNIP3 induces cell death. Herein, we have demonstrated that blockage of hypoxia-induced BNIP3 expression using antisense oligonucleotides against BNIP3 or blockage of BNIP3 function through expression of a mutant form of BNIP3 inhibits hypoxia-induced cell death in human embryonic kidney 293 cells. We have also determined that hypoxia-mediated BNIP3 expression is regulated by the transcription factor, hypoxia-inducible factor-1α (HIF-1α) in human epithelial cell lines. Furthermore, HIF-1α directly binds to a consensus HIF-1α-responsive element (HRE) in the human BNIP3 promoter that upon mutation of this HRE site eliminates the hypoxic responsiveness of the promoter. Since BNIP3 is expressed in hypoxic regions of tumors but fails to induce cell death, we determined whether growth factors block BNIP3-induced cell death. Treatment of the breast cancer cell line MCF-7 cells with epidermal growth factor (EGF) or insulin-like growth factor effectively protected these cells from BNIP3-induced cell death. Furthermore, inhibiting EGF receptor signaling using antibodies against ErbB2 (Herceptin) resulted in increased hypoxia-induced cell death in MCF-7 cells. Taken together, BNIP3 plays a role in hypoxia-induced cell death in human epithelial cells that could be circumvented by growth factor signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bruick RK . (2000). Proc. Natl. Acad. Sci. USA, 97, 9082–9087.
Burris 3rd HA . (2000). Semin. Oncol., 27, 19–23.
Caro J . (2001). High Alt. Med. Biol., 2, 145–154.
Chapman RS, Lourenco PC, Tonner E, Flint DJ, Selbert S, Takeda K, Akira S, Clarke AR and Watson CJ . (1999). Genes Dev., 13, 2604–2616.
Chen G, Cizeau J, Vande Velde C, Park JH, Bozek G, Bolton J, Shi L, Dubik D and Greenberg A . (1999). J. Biol. Chem., 274, 7–10.
Chen G, Ray R, Dubik D, Shi L, Cizeau J, Bleackley RC, Saxena S, Gietz RD and Greenberg AH . (1997). J. Exp. Med., 186, 1975–1983.
Cizeau J, Ray R, Chen G, Gietz RD and Greenberg AH . (2000). Oncogene, 19, 5453–5463.
Clawson TF, Vannucci SJ, Wang GM, Seaman LB, Yang XL and Lee WH . (1999). Biol. Signal. Recept., 8, 281–293.
Farooq M, Kim Y, Im S, Chung E, Hwang S, Sohn M, Kim M and Kim J . (2001). Exp. Mol. Med., 33, 169–173.
Furstenberger G and Senn HJ . (2002). Lancet Oncol., 3, 298–302.
Giatromanolaki A and Harris AL . (2001). Anticancer Res., 21, 4317–4324.
Gibson EM, Henson ES, Haney N, Villanueva J and Gibson SB . (2002). Cancer Res., 62, 488–496.
Gibson S, Tu S, Oyer R, Anderson SM and Johnson GL . (1999). J. Biol. Chem., 274, 17612–17618.
Goonewardene TI, Sowter HM and Harris AL . (2002). Microsc. Res. Technol., 59, 41–48.
Guo K, Searfoss G, Krolikowski D, Pagnoni M, Franks C, Clark K, Yu KT, Jaye M and Ivashchenko Y . (2001). Cell Death Differ., 8, 367–376.
Han BH and Holtzman DM . (2000). J. Neurosci., 20, 5775–5781.
Harris AL . (2002). Nat. Rev. Cancer, 2, 38–47.
Hellermann JP, Jacobsen SJ, Gersh BJ, Rodeheffer RJ, Reeder GS and Roger VL . (2002). Am. J. Med., 113, 324–330.
Hochachka PW, Rupert JL, Goldenberg L, Gleave M and Kozlowski P . (2002). Bioessays, 24, 749–757.
Hon WC, Wilson MI, Harlos K, Claridge TD, Schofield CJ, Pugh CW, Maxwell PH, Ratcliffe PJ, Stuart DI and Jones EY . (2002). Nature, 417, 975–978.
Hung MC and Lau YK . (1999). Semin. Oncol., 26, 51–59.
Izumi Y, Xu L, di Tomaso E, Fukumura D and Jain RK . (2002). Nature, 416, 279–280.
Jin KL, Mao XO and Greenberg DA . (2000). Proc. Natl. Acad. Sci. USA, 97, 10242–10247.
Kim JY, Cho JJ, Ha J and Park JH . (2002). Arch. Biochem. Biophys., 398, 147–152.
Knowles HJ and Harris AL . (2001). Breast Cancer Res., 3, 318–322.
Koukourakis MI . (2001). Anticancer Res., 21, 4285–4300.
Kubasiak LA, Hernandez OM, Bishopric NH and Webster KA . (2002). Proc. Natl. Acad. Sci. USA, 99, 12825–12830.
Kulik G, Klippel A and Weber MJ . (1997). Mol. Cell. Biol., 17, 1595–1606.
Kumar CC . (1998). Oncogene, 17, 1365–1373.
Leek RD, Talks KL, Pezzella F, Turley H, Campo L, Brown NS, Bicknell R, Taylor M, Gatter KC and Harris AL . (2002). Cancer Res., 62, 1326–1329.
Miura M and Yaun J . (2000). Methods Enzymol., 322, 480–492.
Mizutani A, Furukawa T, Adachi Y, Ikehara S and Taketani S . (2002). J. Biol. Chem., 277, 15851–15858.
Mrsic M, Grgic M, Budisic Z, Podolski P, Bogdanic V, Labar B, Jakic-Razumovic J, Restek-Samarzija N and Gosev M . (2001). Ann. Oncol., 12, S95–6.
Piret J, Mottet D, Raes M and Michiels C . (2002). Biochem. Pharmacol., 64, 889.
Ray R, Chen G, Vande Velde C, Cizeau J, Park JH, Reed JC, Gietz RD and Greenberg AH . (2000). J. Biol. Chem., 275, 1439–1448.
Regula KM, Ens K and Kirshenbaum LA . (2002). Circ. Res., 91, 226–231.
Sang N, Fang J, Srinivas V, Leshchinsky I and Caro J . (2002). Mol. Cell. Biol., 22, 2984–2992.
Seigel GM, Chiu L and Paxhia A . (2000). Mol. Vis., 6, 157–163.
Semenza G . (2002a). Biochem. Pharmacol., 64, 993Semenza (2002a). Please provide end page number.–998.
Semenza GL . (2002b). Trends Mol. Med., 8, S62–S67.
Sowter HM, Ratcliffe PJ, Watson P, Greenberg AH and Harris AL . (2001). Cancer Res., 61, 6669–6673.
Stebbing J, Copson E and O’Reilly S . (2000). Cancer Treat. Rev., 26, 287–290.
Vande Velde C, Cizeau J, Dubik D, Alimonti J, Brown T, Israels S, Hakem R and Greenberg AH . (2000). Mol. Cell. Biol., 20, 5454–5468.
Yasuda M, Theodorakis P, Subramanian T and Chinnadurai G . (1998). J. Biol. Chem., 273, 12415–12421.
Yu JL, Rak JW, Coomber BL, Hicklin DJ and Kerbel RS . (2002). Science, 295, 1526–1528.
Acknowledgements
We dedicate this manuscript in the memory of Dr Arnold Greenberg who was a mentor and friend. Dr A Greenberg initiated the project in collaboration with Dr Gibson. This work is supported by Canadian Institutes of Health Research operating grant. L Kirshenbaum is a Canadian Research Chair in Molecular Cardiology. S Kothari is supported by a studentship from the Manitoba Health Research Council. J Cizeau is a recipient of the Shaw Communications/Lymphoma Society of Canada Post-doctoral Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kothari, S., Cizeau, J., McMillan-Ward, E. et al. BNIP3 plays a role in hypoxic cell death in human epithelial cells that is inhibited by growth factors EGF and IGF. Oncogene 22, 4734–4744 (2003). https://doi.org/10.1038/sj.onc.1206666
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206666
Keywords
This article is cited by
-
The ER membrane protein complex restricts mitophagy by controlling BNIP3 turnover
The EMBO Journal (2023)
-
Reinventing the Penumbra — the Emerging Clockwork of a Multi-modal Mechanistic Paradigm
Translational Stroke Research (2023)
-
Tetramethylpyrazine reverses high-glucose induced hypoxic effects by negatively regulating HIF-1α induced BNIP3 expression to ameliorate H9c2 cardiomyoblast apoptosis
Nutrition & Metabolism (2020)
-
Oroxylin A reverses hypoxia-induced cisplatin resistance through inhibiting HIF-1α mediated XPC transcription
Oncogene (2020)
-
Testosterone induces renal tubular epithelial cell death through the HIF-1α/BNIP3 pathway
Journal of Translational Medicine (2019)