Abstract
Purpose
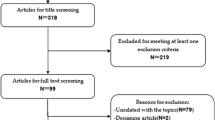
This study compared the efficacy and toxicities of cetuximab combined with chemotherapy versus chemotherapy for patients with metastatic colorectal cancer (mCRC). The influence of KRAS mutation status on the outcomes was also investigated.
Methods
Literature retrieval, trials selection and assessment, data collection, and statistical analysis were performed according to the Cochrane Handbook 5.0.2. The outcome measures were tumor response rate, progression-free survival, overall survival, and adverse effects.
Results
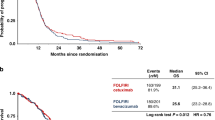
Four randomized controlled trials, comprising totally 2,912 patients, were included. Meta-analysis showed higher response rate (RR 1.93; 95% CI, 1.14–3.26) and significant improvement in progression-free survival (PFS; HR 0.80; 95% CI, 0.67–0.95) in cetuximab-chemotherapy groups versus chemotherapy groups. There was no significant difference between the two treatment groups regarding overall survival (OS; HR 0.95; 95% CI, 0.87–1.05). In wild-type KRAS patients, treatment with cetuximab plus chemotherapy significantly increased response rate (RR 1.44; 95% CI, 1.20–1.73) and improved PFS (HR 0.64; 95% CI, 0.50–0.84), as compared with chemotherapy groups, but not for OS (HR 0.84; 95% CI, 0.64–1.11). In mutant KRAS patients, there was no significant difference between those treated with cetuximab plus chemotherapy and those with chemotherapy alone regarding response rate (RR 0.81; 95% CI, 0.61–1.08), PFS (HR 1.37; 95% CI, 0.81–2.31), and OS (HR 1.03; 95% CI, 0.74–1.44). The risk of grade 3/4 rash, diarrhea, neutropenia, and fatigue was significantly increased in cetuximab combination groups as compared with chemotherapy groups.
Conclusions
The use of cetuximab in addition to chemotherapy was a valid alternative for patients with mCRC. Benefit of cetuximab was only limited to patients with wild-type KRAS tumors.



Similar content being viewed by others
References
Engstrom PF, Arnoletti JP, Benson AB 3rd, Chen YJ, Choti MA, Cooper HS, Covey A, Dilawari RA, Early DS, Enzinger PC, et al (2009) NCCN Clinical Practice Guidelines in Oncology Colon Cancer V.2.2009. National Comprehensive Cancer Network Inc, Washington
American Cancer Society (2009) Cancer facts and figures. http://www.cancer.org/acs/groups/content/@nho/documents/document/500809webpdf.pdf. Accessed Oct 2010
Meyerhardt JA, Mayer RJ (2005) Systemic therapy for colorectal cancer. N Engl J Med 352:476–487
Golfinopoulos V, Salanti G, Pavlidis N, Ioannidis JP (2007) Survival and disease-progression benefits with treatment regimens for advanced colorectal cancer: a meta-analysis. Lancet Oncol 8:898–911
Claire K, Jin C (2007) Chemotherapy in metastatic colorectal cancer. Surg Oncol 16:65–70
Peeters M, Price T, Laetem JLV (2009) Anti-epidermal growth factor receptor monotherapy in the treatment of metastatic colorectal cancer: where are we today? Oncologist 14:29–39
Baselga J, Arteaga CL (2005) Critical update and emerging trends in epidermal growth factor receptor targeting in cancer. J Clin Oncol 23:2445–2459
Van Cutsem E, Köhne CH, Hitre E, Zaluski J, Chang Chien CR, Makhson A, D’Haens G, Pintér T, Lim R, Bodoky G et al (2009) Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N Engl J Med 360:1408–1417
US Food and Drug Administration (2004) ERBITUX® (cetuximab) Solution for intravenous infusion. Initial US. http://www.accessdata.fda.gov/drugsatfda_docs/label/2009/125084s167lbl.pdf. Accessed Sept 2010
Allegra CJ, Jessup JM, Somerfield MR, Hamilton SR, Hammond EH, Hayes DF, McAllister PK, Morton RF, Schilsky RL (2009) American Society of Clinical Oncology provisional clinical opinion: testing for KRAS gene mutations in patients with metastatic colorectal carcinoma to predict response to anti-epidermal growth factor receptor monoclonal antibody therapy. J Clin Oncol 27:2091–2096
European Medicines Agency (2010) Summary of product characteristics—Erbitux. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000558/WC500029119.pdf. Accessed Oct 2010
Bokemeyer C, Bondarenko I, Makhson A, Hartmann JT, Aparicio J, de Braud F, Donea S, Ludwig H, Schuch G, Stroh C et al (2009) Fluorouracil, leucovorin, and oxaliplatin with and without cetuximab in the first-line treatment of metastatic colorectal cancer. J Clin Oncol 27:663–671
Sobrero AF, Maurel J, Fehrenbacher L, Scheithauer W, Abubakr YA, Lutz MP, Vega-Villegas ME, Eng C, Steinhauer EU, Prausova J et al (2008) EPIC: phase III trial of cetuximab plus irinotecan after fluoropyrimidine and oxaliplatin failure in patients with metastatic colorectal cancer. J Clin Oncol 26:2311–2319
Borner M, Koeberle D, Von Moos R, Saletti P, Rauch D, Hess V, Trojan A, Helbling D, Pestalozzi B, Caspar C et al (2008) Adding cetuximab to capecitabine plus oxaliplatin (XELOX) in first-line treatment of metastatic colorectal cancer: a randomized phase II trial of the Swiss Group for Clinical Cancer Research SAKK. Ann Oncol 19:1288–1292
Baselga J, Norton L, Masui H, Pandiella A, Coplan K, Miller WH Jr, Mendelsohn J (2008) Antitumor effects of doxorubicin in combination with anti-epidermal growth factor receptor monoclonal antibodies. J Natl Cancer Inst 85:1327–1333
Bruns CJ, Harbison MT, Davis DW, Portera CA, Tsan R, McConkey DJ, Evans DB, Abbruzzese JL, Hicklin DJ, Radinsky R (2000) Epidermal growth factor receptor blockade with C225 plus gemcitabine results in regression of human pancreatic carcinoma growing orthotopically in nude mice by antiangiogenic mechanisms. Clin Cancer Res 6:1936–1948
Inoue K, Slaton JW, Perrotte P, Davis DW, Bruns CJ, Hicklin DJ, McConkey DJ, Sweeney P, Radinsky R, Dinney CP (2000) Paclitaxel enhances the effects of the anti-epidermal growth factor receptor monoclonal antibody ImClone C225 in mice with metastatic human bladder transitional cell carcinoma. Clin Cancer Res 6:4874–4884
Schilsky RL (2002) End points in cancer clinical trials and the drug approval process. Clin Cancer Res 8:935–938
Benvenuti S, Sartore-Bianchi A, Di Nicolantonio F, Zanon C, Moroni M, Veronese S, Siena S, Bardelli A (2007) Oncogenic activation of the RAS/RAF signaling pathway impairs the response of metastatic colorectal cancer to anti-epidermal growth factor receptor antibody therapies. Canc Res 67:2643–2648
Karapetis CS, Khambata-Ford S, Jonker DJ, O’Callaghan CJ, Tu D, Tebbutt NC, Simes RJ, Chalchal H, Shapiro JD, Robitaille S et al (2008) K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N Engl J Med 359:1757–1765
Van Cutsem E, Peeters M, Siena S, Humblet Y, Hendlisz A, Neyns B, Canon JL, Van Laethem JL, Maurel J, Richardson G et al (2007) Open-label phase III trial of panitumumab plus best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. J Clin Oncol 25:1658–1664
Arkenau HT, Arnold D, Cassidy J, Diaz-Rubio E, Douillard JY, Hochster H, Martoni A, Grothey A, Hinke A, Schmiegel W et al (2008) Efficacy of oxaliplatin plus capecitabine of infusional fluorouracil/leucovorin in patients with metastatic colorectal cancer: a pooled analysis of randomized trials. J Clin Oncol 26:5910–5917
Jonker DJ, O’Callaghan CJ, Karapetis CS, Zalcberg JR, Tu D, Au HJ, Berry SR, Krahn M, Price T, Simes RJ et al (2007) Cetuximab for the treatment of colorectal cancer. N Engl J Med 357:2040–2048
Wang T, Luo L, Zhou Q, Wu T (2008) Irinotecan versus oxaliplatin in combination with 5-FU/LV for advanced colorectal cancer: a systematic review. Chinese Journal of Evidence-Based Medicine 8:36–41
Ocvirk J, Brodowicz T, Wrba F, Ciuleanu TE, Kurteva G, Beslija S, Koza I, Pápai Z, Messinger D, Yilmaz U et al (2010) Cetuximab plus FOLFOX6 or FOLFIRI in metastatic colorectal cancer: CECOG trial. World J Gastroenterol 16:3133–3143
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, L., Ma, L. & Zhou, Q. Overall and KRAS-specific results of combined cetuximab treatment and chemotherapy for metastatic colorectal cancer: a meta-analysis. Int J Colorectal Dis 26, 1025–1033 (2011). https://doi.org/10.1007/s00384-011-1197-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-011-1197-5