Abstract
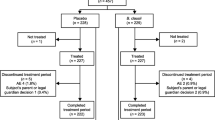
Probiotics have been successfully used for the treatment of acute diarrhea in children and this effect depends on the strains and dose. The aim of this study was to assess the effect of a synbiotic mixture on the duration of diarrhea and the length of hospital stay in children with acute watery diarrhea. This is a prospective randomized, multicenter single blinded clinical trial in hospitalized children with acute watery diarrhea. All children were treated with conventional hydration therapy with or without a daily dose of a synbiotic (2.5 × 109 CFU live bacteria including Lactobacillus acidophilus, Lactobacillus rhamnosus, Bifidobacterium bifidum, Bifidobacterium longum, Enterococcus faecium, and 625 mg fructooligosaccharide) for 5 days. The primary endpoint was duration of diarrhea and duration of hospitalization was the secondary endpoint. Among 209 eligible children, 113 received the synbiotic mixture and 96 served as a control. The duration of diarrhea was significantly shorter (∼36 h) in children receiving the synbiotic group than the controls (77.9 ± 30.5 vs. 114.6 ± 37.4 h, p < 0.0001). The duration of hospitalization was shorter in children receiving the synbiotic group (4.94 ± 1.7 vs. 5.77 ± 1.97 days, p = 0.002). The effect of synbiotic mixture on diarrhea started after 24th hours and stool frequency significantly decreased after 24th and 48th hours. The percentage of diarrhea-free children is significantly higher in synbiotic group at 48th and 72nd hours of synbiotic group. In conclusion, this study showed a reduction in diarrhea duration by approximately 36 h and a reduction in the duration of hospitalization with approximately 1 day in children with acute diarrhea with this synbiotic mixture.


Similar content being viewed by others
References
Allen SJ, Martinez EG, Gregorio GV, Dans LF (2010) Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst Rev 10:CD003048
Canani RB, Cirillo P, Terrin G, Cesarano L, Spagnuolo MI, De Vincenzo A, Albano F, Passariello A, De Marco G, Manguso F, Guarino A (2007) Probiotics for treatment of acute diarrhoea in children: randomised clinical trial of five different preparations. BMJ 335:340
Cha MJ, Chung MJ, Kim JE, Lee KO, Ha NJ (2011) Comparison of dual coated (Duolac) and uncoated lactic acid bacteria from potential probiotics. Biotechnol Biotechnol 25:2489–2493
Chen CC, Kong MS, Lai MW, Chao HC, Chang KW, Chen SY, Huang YC, Chiu CH, Li WC, Lin PY, Chen CJ, Li TY (2010) Probiotics have clinical, microbiologic, and immunologic efficacy in acute infectious diarrhea. Pediatr Infect Dis J 29:135–138
Cheng AC, McDonald JR, Thielman NM (2005) Infectious diarrhea in developed and developing countries. J Clin Gastroenterol 39:757–773
Dinleyici EC, Eren M, Ozen M, Yargic ZA, Vandenplas Y (2012) Effectiveness and safety of Saccharomyces boulardii for acute infectious diarrhea. Expert Opin Biol Ther 12:395–410
Floch MH, Walker WA, Madsen K et al (2011) Recommendations for probiotic use—2011 update. J Clin Gastroenterol 45 Suppl:S168–S171
Gadewar S, Fasano A (2005) Current concepts in the evaluation, diagnosis and management of acute infectious diarrhea. Curr Opin Pharmacol 5:559–565
Grandy G, Medina M, Soria R, Terán CG, Araya M (2010) Probiotics in the treatment of acute rotavirus diarrhoea. A randomized, double-blind, controlled trial using two different probiotic preparations in Bolivian children. BMC Infect Dis 10:253
Guandalini S (2011) Probiotics for prevention and treatment of diarrhea. J Clin Gastroenterol 45(Suppl):S149–S153
Guarino A, Albano F, Ashkenazi S et al (2008) European Society for Paediatric Gastroenterology, Hepatology, and Nutrition/European Society for Paediatric Infectious Diseases evidence-based guidelines for the management of acute gastroenteritis in children in Europe. J Paediatr Gastroenterol Nutr 46(Suppl 2):S81–S122
Guarner F, Khan AG, Garisch J, Eliakim R, Gangl A, Thomson A, Krabshuis J, Lemair T, Kaufmann P, de Paula JA, Fedorak R, Shanahan F, Sanders ME, Szajewska H, Ramakrishna BS, Karakan T, Kim N, Review Team; Invited outside experts (2012) World Gastroenterology Organisation Global Guidelines: probiotics and prebiotics. J Clin Gastroenterol 46:468–481
Hoekstra JH, Szajewska H, Zikri MA, Micetic-Turk D, Weizman Z, Papadopoulou A, Guarino A, Dias JA, Oostvogels B (2004) Oral rehydration solution containing a mixture of non-digestible carbohydrates in the treatment of acute diarrhea: a multicenter randomized placebo controlled study on behalf of the ESPGHAN working group on intestinal infections. J Pediatr Gastroenterol Nutr 39:239–245
O'Ryan M, Lucero Y, O'Ryan-Soriano MA, Ashkenazi S (2010) An update on management of severe acute infectious gastroenteritis in children. Expert Rev Anti Infect Ther 8:671–682
Passariello A, Terrin G, De Marco G, Cecere G, Ruotolo S, Marino A, Cosenza L, Tardi M, Nocerino R, Berni Canani R (2011) Efficacy of a new hypotonic oral rehydration solution containing zinc and prebiotics in the treatment of childhood acute diarrhea: a randomized controlled trial. J Pediatr 158:288–289
Vandenplas Y, De Hert SG, PROBIOTICAL-study group (2011) Randomised clinical trial: the synbiotic food supplement Probiotical vs. placebo for acute gastroenteritis in children. Aliment Pharmacol Ther 34(8):862–867
Vandenplas Y, Salvatore S, Vieira M, Devreker T, Hauser B (2007) Probiotics in infectious diarrhoea in children: are they indicated? Eur J Pediatr 166:1211–1218
Vandenplas Y, Veereman-Wauters G, De Greef E et al (2011) Probiotics and prebiotics in prevention and treatment of diseases in infants and children. J Pediatr (Rio J) 87:292–300
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dinleyici, E.C., Dalgic, N., Guven, S. et al. The effect of a multispecies synbiotic mixture on the duration of diarrhea and length of hospital stay in children with acute diarrhea in Turkey: Single blinded randomized study. Eur J Pediatr 172, 459–464 (2013). https://doi.org/10.1007/s00431-012-1903-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-012-1903-5